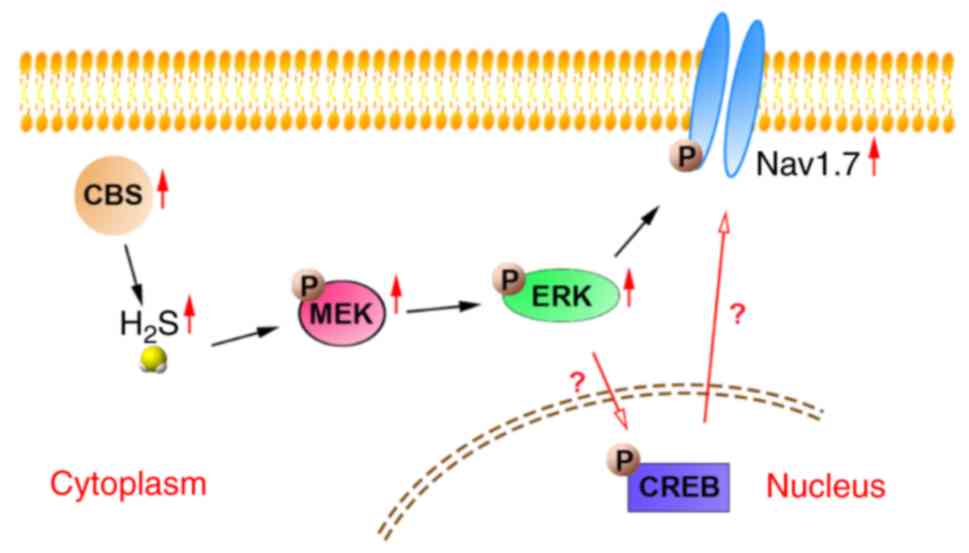
Lending credence to the involvement of Nav channel aberrations in neuropsychiatric disorders, Nav channel blockers are also commonly used as adjunct therapies for the treatment of bipolar disorder, anxiety, and schizophrenia. Given their primacy in modulating neuronal excitability, it is unsurprising that aberrant activity of these isoforms has been implicated in the etiologies of numerous neurologic and neurodevelopmental disorders, including epilepsy, migraines, and autism. Of the nine different pore-forming α-subunits that have been described (Nav1.1–1.9), the Nav1.1–1.3 and 1.6 isoforms are the primary Nav channels expressed in the central nervous system (CNS). In excitable cells, voltage-gated sodium (Nav) channels are the primary molecular determinants of the generation and conduction of action potentials. Overall, we have identified a negative and positive allosteric modulator of Nav1.6, both of which could serve as scaffolds for the development of target-selective neurotherapeutics. Subsequent functional evaluation using whole-cell patch-clamp electrophysiology confirmed their inverse activities, with 12 and 19 reducing and increasing Nav1.6-mediated transient current densities, respectively. Conversely, addition of hydrophobic protective groups to 6 followed by addition of an N-terminal benzoyl substituent ( 19) produced a potentiator of FGF14:Nav1.6 complex assembly. Addition of hydrophobic protective groups to 6 and truncation to a tripeptide ( 12) produced a potent inhibitor of FGF14:Nav1.6 complex assembly. 
Here, peptidomimetics derived from EYYV ( 6) were designed, synthesized, and pharmacologically evaluated to develop probes with improved potency. The tetrapeptide Glu-Tyr-Tyr-Val (EYYV) mimics surface residues of FGF14 at the β8–β9 loop, a structural region previously implicated in its binding to Nav1.6. Thus, peptides derived from FGF14 might exert modulatory actions on the FGF14:Nav1.6 complex that are functionally relevant. One example of this pathophysiology is mediated by dysfunction of the PPI between Nav1.6 and its regulatory protein fibroblast growth factor 14 (FGF14). Disruption of protein:protein interactions (PPIs) that regulate the function of voltage-gated Na + (Nav) channels leads to neural circuitry aberrations that have been implicated in numerous channelopathies.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
